Bengaluru, Karnataka, India: May 7, 2026:
Biocon Limited (BSE code: 532523, NSE: BIOCON), an innovation-led global biopharmaceuticals company, today announced its consolidated financial results for the fiscal fourth quarter and the financial year ended March 31, 2026.
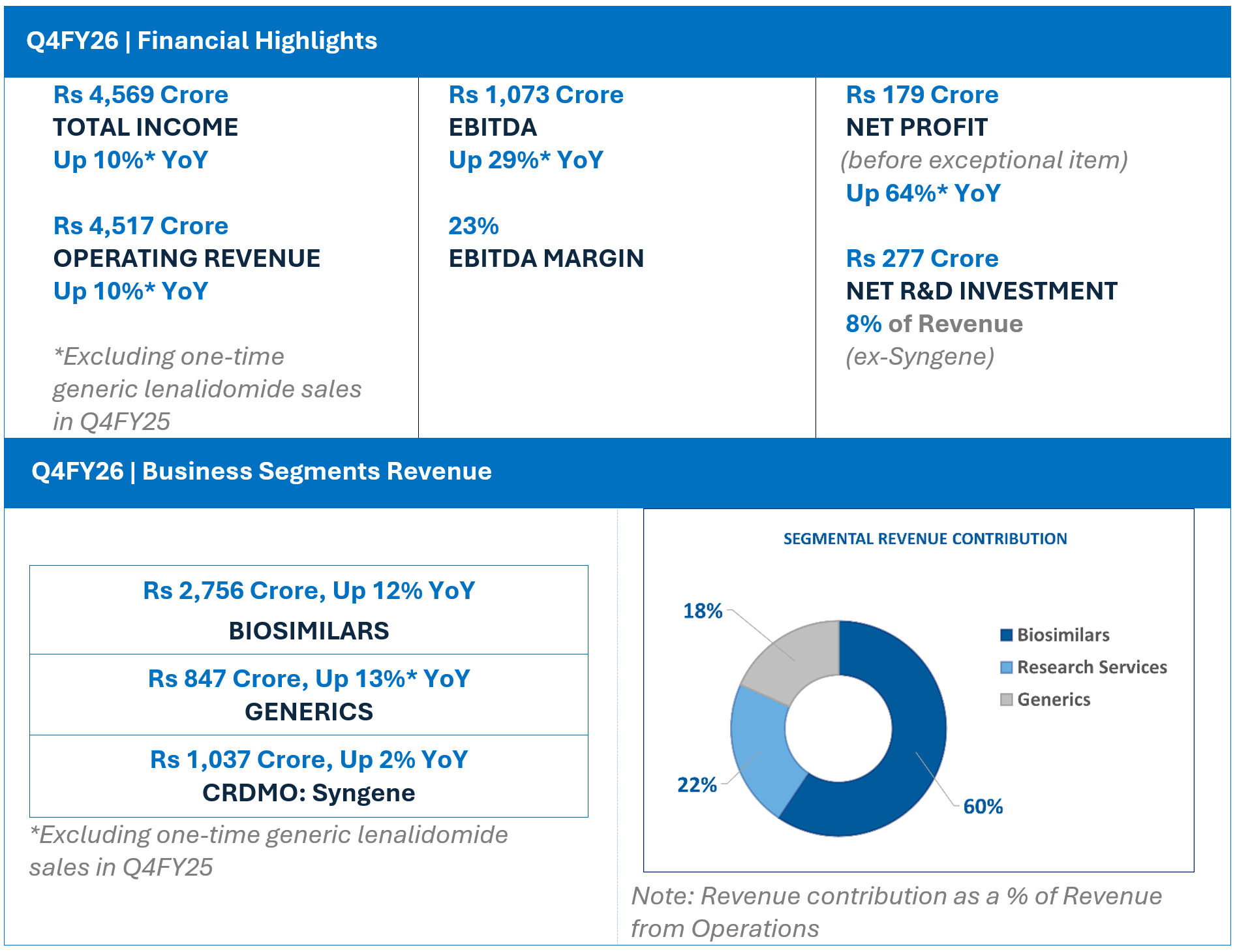
Biocon Q4FY26 Total Income at Rs 4,569 Cr EBITDA at Rs 1,073 Cr; Net Profit (before exceptional items) at Rs 179 Cr FY26 Total Income at Rs 17,270 Cr; EBITDA at Rs 3,798 Cr; Net Profit (before exceptional items) at Rs 436 Cr
-
Thu, 07-May-2026
- Posted by: BIOCON

Leadership Comments
“Biocon closed FY26 on a strong note despite a complex geopolitical environment. We delivered margin expansion along with 13% YoY growth in operating revenue, excluding the one-time impact of generic lenalidomide sales last year. This performance reflects the resilience of our business and disciplined execution through a pivotal year of integration. We have created a unified biopharma entity by integrating biosimilars business with generics formulations and APIs business. The combined business has a stronger balance sheet, improved leverage metrics and a more global commercial footprint. We are now focused on profitability enhancement and long-term value creation.”
– Kiran Mazumdar-Shaw, Executive Chairperson, Biocon Limited
“FY26 was a pivotal year for Biocon, marked by the successful integration of our biosimilars and generics business to create one unified biopharmaceutical enterprise with greater scale, capability, and reach. Even as we integrated the businesses, Biocon delivered ~200 basis points of EBITDA margin expansion at a consolidated level on a like-to-like basis, driven by an improved product mix and operational excellence. Our biosimilars business recorded 16% YoY growth in revenue and 40% increase in EBITDA, on a like-to-like basis. Growth was led by strong performance in advanced markets and key tender wins across emerging markets. With the integration now complete, we shift gears in fiscal 2027 from the “Preserve” phase of our strategy to “Consolidate”, as we set up the business for sustainable growth.”
– Shreehas Tambe, CEO & Managing Director, Biocon Limited
“Syngene’s full-year revenue from operations grew 3%, and with an EBITDA margin of 25%, performance was in line with our revised full-year guidance. The overall numbers reflect the specific impact from a single large-molecule biologics client, with the underlying business showing steady momentum. During the year, we continued to invest in new capabilities and emerging modalities such as peptides and ADCs, further strengthening our integrated offering and positioning us for long-term growth.”
– Peter Bains, CEO & Managing Director, Syngene International Limited
FINANCIAL HIGHLIGHTS (CONSOLIDATED): Q4FY26 & Full Year FY26
In Rs Crore
| Particulars | Q4FY26 | Q4FY25 (reported) | Q4 FY25 (adjusted) | YoY (%) | FY26 | FY25
(reported) |
FY25
(adjusted) |
YoY (%) |
| INCOME | ||||||||
| Biosimilars | 2,756 | 2,463 | 2,463 | 12 | 10,431 | 9,017 | 9,017 | 16 |
| Generics | 847 | 1048 | 748 | 13* | 3,168 | 3,017 | 2,717 | 17* |
| CRDMO/ Research Services | 1,037 | 1,018 | 1,018 | 2 | 3,739 | 3,642 | 3,642 | 3 |
| Inter-segment | (122) | (112) | (112) | 9 | (411) | (416) | (416) | (1) |
| Revenue from operations# | 4,517 | 4,417 | 4,117 | 10* | 16,927 | 15,262 | 14,962 | 13* |
| Other income | 53 | 37 | 37 | 42 | 343 | 1,208 | 151 | 1271 |
| Total Income | 4,569 | 4,454 | 4,154 | 10* | 17,270 | 16,470 | 15,113 | 14@ |
| Net R&D Expenses | 277 | 231 | 231 | 20 | 982 | 859 | 859 | 14 |
| EBITDA | 1,073 | 1,115 | 834 | 29* | 3,798 | 4,374 | 3,036 | 25@ |
| EBITDA Margins | 23% | 25% | 20% | 22% | 27% | 20% | ||
| PBT (before Exceptional Items^) | 328 | 466 | 185 | 77* | 851 | 1,790 | 452 | 88@ |
| Net Profit (before Exceptional Items^^) | 179 | 333 | 109 | 64* | 436 | 981 | 103 | 323@ |
| Net Profit (Reported) | 126 | 344 | 120 | 5* | 386 | 1,013 | 135 | 186@ |
Figures above are rounded off to the nearest Crore; % based on absolute numbers.
Notes to financials above:
#Revenue from operations includes licensing income
1Adjusted for BFI Divestment Gain of Rs 1,057 Cr
*Excluding one-time generic lenalidomide sales in Q4FY25
@Excluding one-time generic lenalidomide sales in Q4FY25 & adjusted for BFI divestment gain
^Exceptional items during Q4FY26 and FY26 amount to Rs (80) crore and Rs (403) crore, respectively
^^Net of tax and minority interest, exceptional items during Q4FY26 and FY26 amounted to Rs. (53) crore and Rs (50) crore respectively, resulting in a Net Profit of Rs 126 crore and Rs 386 crore, respectively
Financial Commentary: Q4FY26
- Operating Revenue at Rs 4,517 crore, up 10% year-on-year (YoY), after adjusting for one-time generic Lenalidomide sales in Q4FY25.
- EBITDA at Rs 1,073 crore. Adjusted for generic Lenalidomide sales, EBITDA was up 29% YoY, with a margin of 23%.
- Net Profit (before exceptional items) is Rs 179 crore.
Financial Commentary: FY26
- Operating Revenue at Rs 16,927 crore, up 13% year-on-year (YoY), after adjusting for one-time generic Lenalidomide sales in FY25.
- EBITDA at Rs 3,798 crore. Adjusted for generic Lenalidomide sales and for BFI divestment gain, EBITDA was up 25% YoY.
- EBITDA margin at 22%, up 200 basis points, on a like-to-like basis.
- Net Profit (before exceptional items) is Rs 436 crore.
Dividend for FY25
The Board of Directors has recommended a final dividend of Re 0.50 per share at the rate of 10% of the face value of the share, for the financial year ending March 31, 2026.
Board Updates
Based on the recommendations of the Nomination & Remuneration Committee, the Board has recommended for approval of the shareholders the appointment of Rajiv Malik, Daniel Bradbury, Peter Piot, Arun Chandavarkar and Nivruti Rai as Independent Directors, each with effect from August 1, 2026, until the conclusion of the Company’s 50th AGM to be held in the year 2028. The Board also recommended the appointment of Thomas Roberts as a Non-Executive, Non-Independent Director with effect from August 1, 2026.
All the above appointed Board members have served on the Board of Biocon Biologics.
Management Updates
Shreehas Tambe has taken charge as CEO & Managing Director of Biocon Limited on April 1, 2026, with Kedar Upadhye appointed as Chief Financial Officer.
Awards and Recognitions
- Kiran Mazumdar-Shaw conferred Honorary Doctor of Philosophy in Applied Life Sciences by the Keck Graduate Institute.
- Kiran Mazumdar-Shaw received an Honorary Fellowship of the UK Academy of Medical Sciences.
- Kiran Mazumdar-Shaw conferred with SAHIT Global Icon Award.
- Shreehas Tambe recognized as one of the Most Promising Business Leaders of Asia 2026.
Sustainability / ESG
- Biocon included in the S&P Global Sustainability Yearbook 2026 for the fourth consecutive year.
- Biocon ranked among Businessworld’s Top 20 Most Sustainable Companies in India, securing 3rd position in the pharma sector.
- Syngene included in the S&P Global Sustainability Yearbook 2026; was also recognized as an ‘Industry Mover’.
Business Highlights
BIOSIMILARS
- Q4FY26 Revenue from Operations at Rs 2,756 crore, up 12% YoY
- Q4FY26 EBITDA at Rs 720 crore, up 33%; representing EBITDA Margin of 26%
- FY26 Revenue from Operations at Rs 10,431 crore, up 16% YoY
- FY26 EBITDA at Rs 2,751 crore, up 40% YoY, adjusted for BFI divestment gain; representing EBITDA Margin of 26%
- Served 6.5+ Million Patients (MAT March 2026 basis) ##
##12-month moving annual patient population (April 2025 to March 2026)
Business Performance
The Biosimilars business remained the core growth driver. Operating revenue for Q4FY26 stood at Rs 2,756 crore, up 12% YoY, driven primarily by performance in the Advanced Markets. Recent launches scaled up supporting growth and operating leverage. EBITDA for the quarter stood at Rs 720 crore, up 33% YoY, translating into an EBITDA margin of 26%. Margins in this segment reflected the benefits of improvement in revenue mix as well as operating leverage. R&D investments for the quarter stood at 7% of revenues.
For FY26, Operating revenue and EBITDA grew at 16% and 40% respectively, on a like-to-like basis.
The Company launched Bosaya™ and Aukelso™, the Denosumab biosimilars to Prolia® and Xgeva®, marking a strategic expansion of its biosimilars portfolio in the U.S. It also obtained key approvals, including Health Canada approvals for Denosumab biosimilars, Bosaya™ and Vevzuo™.
GENERICS: APIs & Generic Formulations
- Q4FY26 Revenue from Operations at Rs 847 Crore, up 13% YoY, after adjusting for one-time generic Lenalidomide supplies in Q4FY25.
- Q4FY26 EBITDA at Rs 75 crore. EBITDA margin at 8%, up ~300 basis points QoQ, driven by higher volumes and operating leverage.
- FY26 Revenue from Operations at Rs 3,168 Crore, up 17% YoY, adjusted for generic lenalidomide sales.
Business Performance
Operating revenue stood at Rs 847 crore. Adjusted for one-time generic Lenalidomide supplies in Q4FY25, revenues grew 13% YoY, driven by generic Liraglutide sales in Europe. EBITDA stood at Rs 75 crore with an 8% margin, improving nearly 300 basis points QoQ.
During the quarter, the Company secured U.S. FDA approval for generic Liraglutide, covering both diabetes and weight management indications. For FY26, Generics revenues grew 17% YoY on a like-to-like basis.
CRDMO: Syngene
- Q4FY26 Revenue from Operations at Rs 1,037 Crore, up 2% YoY
- FY26 Revenue from Operations at Rs 3,739 Crore, up 3% YoY
Business Performance
Syngene’s Q4FY26 operating revenue at Rs 1,037 crore were up 2% YoY and 13% QoQ.
FY26 operating revenues stood at Rs 3,739 crore, reflecting a 3% YoY increase. EBITDA margin for the year was 25%, in line with revised full‑year guidance.
Syngene completed 14 client and regulatory audits during the quarter, bringing the full-year total to 85.
Note: Biocon in Q1FY26 renamed its Research Services business segment as CRDMO to represent Syngene’s business model of a CRO + CMO.
About Biocon Limited
Biocon Limited (BSE: 532523, NSE: BIOCON) is a global biopharmaceutical company driven by its purpose to provide affordable, life-changing medicines to patients worldwide. Headquartered in Bengaluru, India, Biocon addresses some of the world’s most pressing healthcare challenges across chronic and non-communicable diseases by offering both biosimilars and generics at scale across geographies. Through this diversified portfolio, Biocon focuses on areas of high unmet need, spanning key therapy areas including diabetes, oncology, obesity, cardiovascular diseases, immunology, ophthalmology, and bone health. The Company has pioneered several industry firsts that have helped shape the global biosimilars landscape. To date, the company has commercialized 12 biosimilar products and 30+ generic formulations globally. It has robust research and development pipeline of 20+ biosimilar assets, as well as GLP-1 peptides and other complex generics. With an integrated lab-to-patient model, Biocon brings together research and development, manufacturing, and commercial capabilities to ensure reliable and scalable supply of medicines. The company operates in more than 120 countries, supported by seven manufacturing sites, three R&D sites, 18 offices worldwide, and a workforce of over 9,500 employees. Biocon has been included in the S&P Global Sustainability Yearbook 2026 for the fourth consecutive year, underscoring its commitment to sustainable and responsible growth. Website: www.biocon.com Follow us on X: @bioconlimited LinkedIn: Biocon
About Syngene International Ltd.
Syngene International Ltd. (BSE: 539268, NSE: SYNGENE, ISIN: INE 398R01022) is an integrated research, development, and manufacturing services company serving the global pharmaceutical, biotechnology, nutrition, animal health, consumer goods, and specialty chemical sectors. Syngene’s team of over 5,600 scientists brings both deep expertise and the capacity to deliver scientific excellence, robust data security, and world class manufacturing, at speed, to improve time-to-market and lower the cost of innovation. With 2.5+ mn sq. ft of specialized discovery, development, and manufacturing facilities, Syngene works with around 400 global customers across industry segments, including biotech companies pursuing leading-edge science and multinationals such as BMS, GSK, Zoetis, and Merck KGaA. For more details, visit www.syngeneintl.com . For the Company’s latest Environmental, Social, and Governance (ESG) report, visit Syngene ESG Report.
Forward-Looking Statements: Biocon
This press release may include statements of future expectations and other forward-looking statements based on management’s current expectations and beliefs concerning future developments and their potential effects upon Biocon and its subsidiaries/ associates. These forward-looking statements involve known or unknown risks and uncertainties that could cause actual results, performance or events to differ materially from those expressed or implied in such statements. Important factors that could cause actual results to differ materially from our expectations include, amongst other: general economic and business conditions in India and overseas, our ability to successfully implement our strategy, our research and development efforts, our growth and expansion plans and technological changes, changes in the value of the Rupee and other currency changes, changes in the Indian and international interest rates, change in laws and regulations that apply to the Indian and global biotechnology and pharmaceuticals industries, increasing competition in and the conditions of the Indian and global biotechnology and pharmaceuticals industries, changes in political conditions in India and changes in the foreign exchange control regulations in India. Neither Biocon, nor our Directors, or any of our subsidiaries/associates assume any obligation to update any particular forward-looking statement contained in this release.
